 On the other hand, the TME also contains a rich repertoire of tumor-infiltrating immune cells including T- and B-cells, neutrophils, dendritic cells, myeloid-derived suppressor cells, or tumor-associated macrophages, that normally constitute a natural barrier to carcinogenesis ( 22, 24). Among malignant cells, the TME contains tumor cell subclones expressing phenotypic traits that protect them from the hosts immune system and support their ability to invade the extracellular matrix and extravasate ( 22, 28). 
The TME is the ensemble of tumor cells, non-tumor cells including carcinoma-associated fibroblasts and immune cells, extra-cellular matrix as well as blood and lymphatic vessels composing a neoplastic lesion ( 24– 27). In this review, we discuss how both genetic and non-genetic intratumor heterogeneity (ITH) influences the immunogenicity of M-NSCLC, and highlight the importance of integrated genomic, pathologic and immunologic analyses to refine the selection of M-NSCLC patients who may be candidates to treatment with ICIs.Ī key determinant of the response of M-NSCLC patients to ICI therapy is the tumor microenvironment (TME) ( 21– 23). However, many factors contribute to the extent of the response as well as the risk of developing resistance to ICIs in these patients ( 1, 3). Most studies conducted so far have shown that the response to ICIs in M-NSCLC patients is independent of the histological subtype (squamous or non-squamous histology) ( 1, 3). Moreover, there is now evidence that dual blockade of CTLA-4 and PD-1 receptors is sufficient to induce unique cellular responses compared with agents blocking these receptors given alone to M-NSCLC patients ( 20). However, the response to single-agent ICI therapy is not durable, and only a minority of patients have a prolonged benefit ( 2, 9, 14). ICIs have proven to be better tolerated than standard chemotherapy ( 2, 6, 10). The selection of which ICI to use depends on the expression of PD-L1, which can be evaluated using various assays, whose clinical validity has been assessed in numerous clinical trials ( 5, 6, 8– 10, 12– 16). Commonly used ICIs in these patients include the monoclonal antibodies: nivolumab ( 5– 8), pembrolizumab ( 9– 12), durvalumab ( 13), atezolizumab ( 14– 16), and avelumab ( 17), which act by targeting immune checkpoints expressed by tumor infiltrating lymphocytes (TILs)- programmed-death 1 (PD-1)-or expressed by cancer and tumor infiltrating immune cells- programmed-death ligand 1 (PD-L1) ( 18, 19). The availability of immune checkpoint inhibitors (ICIs) has radically changed the management of patients affected by M-NSCLC ( 3). Modulation of interactions between T-cells, antigen-presenting cells, and tumor cells has helped unleash suppressed immune responses and increase the effective elimination of cancer cells ( 1, 2).  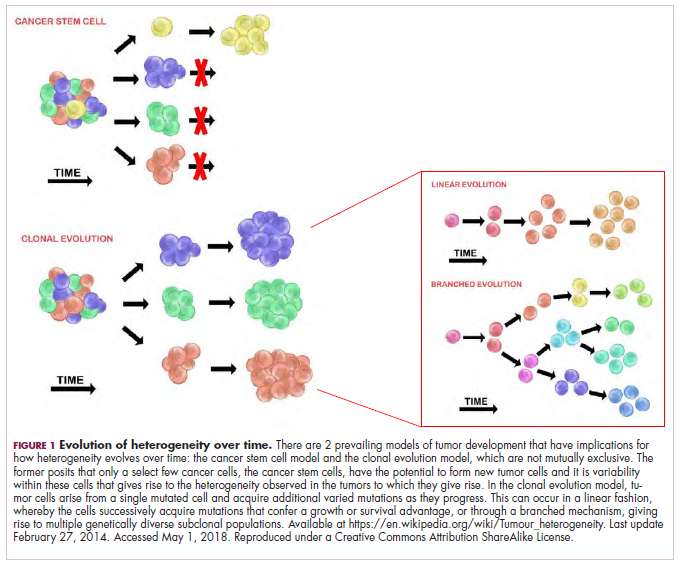
This key observation has paved the way to the development of immunomodulating agents and opened the era of cancer immunotherapy ( 3), which culminated with the assignment of the Nobel Price to James P. By now, it is clear that the immune system plays a pivotal role not only in eradicating the disease in cancer patients, but also in promoting a long-lasting immunity ( 2). Understanding the interactions between the immune system and cancer cells has greatly advanced our knowledge of the mechanisms of tumor growth and progression ( 1). Immune Checkpoint Inhibitors in Metastatic Non-Small Cell Lung Cancer: An Overview 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
